Health
The FDA is trying to shut down his successful cancer treatments. Dr. Stanislaw Burzynski’s patients are outraged

The FDA’s Relentless Persecution Of Dr. Stanislaw Burzynski
Patients and even grand jurors were outraged at the prosecutors going after Burzynski. Readers have asked me to assess the efficacy of his anti-neoplaston therapy using the Kory Scale. Lets do it.
In a recent post, I tried to introduce a conceptual tool which I (as humorously and self-deprecatingly as possible) named after myself. In brief, “The Kory Scale” (TKS) is based on the hypothesis that the more a new or existing medical therapy is attacked in a coordinated, sustained fashion by the medical establishment and media, the more likely the treatment is both highly effective and safe.
This hypothesis is based on the simple fact that the more effective and safe (and cheap and widely available) a therapy is, the more it threatens the massive markets of the bio-pharmaceutical industrial complex. And that industry “don’t play” when their profits are threatened.
Thus, scores on TKS increase proportionally to the degree, breadth, and viciousness of the attacks generated from “the Pharma controlled medical establishment” in response to reports of efficacy of the proposed therapy (i.e. attacks from the AMA, FDA, NIH, DOJ, medical journals, pharma-controlled media, and the military).
Dr. Stanislaw Burzynski and his “Anti-Neoplaston” Therapy
A couple of years ago I watched this shocking documentary about the decades long plight of harassment of Dr. Burzynski by the FDA, the Texas Medical Board, and the National Cancer Institute. I have to admit that it likely became a major influence in my inspiration for The Kory Scale.
Let me just say straight off that I know little about anti-neoplaston therapy outside of what I learned in the documentary. However, I do know professionally that it has been extremely effective and/or curative in some patients while others have not reported responses (welcome to cancer). The exact incidence and magnitude of effectiveness is thus unknown to me but I argue that based on its score on TKS, it must be at least moderately effective and even if the overall incidence of efficacy is low, it can and has led to a considerable number of remarkable documented remissions of advanced cancers, especially in the brain.
Further, through my review of his case, Burzynski never “promised it as a cure” because he knew it did not work in everybody, but he had accumulated enough dramatic responses and full remissions to know it should simply be a treatment option in cancer, but not a cure-all for cancer as there is NO SUCH THING. Pharma knows that reality all too well. For instance, outside of a handful of chemo-sensitive cancers, chemo has an absolutely atrocious record of toxicity and inefficacy in treating cancer, particularly with solid tumors given that on average it merely extends life for varying periods of often just months while incurring significant toxicity and loss of quality of life. For instance:
- Age-adjusted death rates for cancer have remained remarkably stable or have even increased since 1930.
- In the last 15 years, chemo and new cancer therapies have led to an improvement in survival of just 2.4 months.
Another aspect that I believe brought Burzynski even more persecution is that anti-neoplaston treatment is not cheap as it runs approximately $9,000 a month. Apparently it is quite costly to collect and purify the peptides from healthy donors. That price point is likely what made it attractive to those that later tried to “steal” his therapy from him. I also suspect the treatment got even more expensive due to the massive legal bills incurred through the 3 decades of innumerable indictments by “the establishment.” The final blow making his therapy unaffordable to many (which you will learn of below) was the collusion of the health insurance companies in not covering anti-neoplastons.
Now, before we run antineoplastons through the Kory Scale, know that I have since revised the elements on the scale, (adding some novel actions take against Burzynski) while adopting a more categorical than discrete scoring system in assigning points to such actions (point assignments are still personal and arbitrary though).
Version 2.0:
The History Of Dr. Stanislaw Burzynski and Anti-Neoplaston Therapy
A Polish native named Stanislaw Burzynski attended Brooklyn Medical University, where he graduated first in his class at age 24, and then received his PhD in biochemistry the following year. While undergoing his research to acquire his PhD, Dr. Burzynski made a profound discovery. He found a strain of peptides in human blood and urine that had never before been recorded in biomechanical research. As his curiosity in his peptides evolved, he made another profound observation. People with cancer seemed to lack these newly discovered peptides in both their blood and urine, while those who were healthy appeared to have an abundance of these peptides. Dr. Brzezinski wondered if there was a way to chemically extract these peptides from the blood and urine of healthy donors and administer these peptides to those with cancer. Perhaps it could be useful in treating the disease.
Further, from this account on the Wayback machine:
In 1970, three years after graduating from the Medical Academy of Lublin, Burzynski emigrated to the United States and took a position as researcher and associate professor at the Baylor College of Medicine in Houston. With the help of a grant from the National Cancer Institute, Burzynski continued his antineoplaston research. But when a grant renewal was denied in 1976, Burzynski decided that if he wanted to see his theory through to the end, he would have to do it on his own.
But first he had to find out if it was even legal to manufacture, sell and administer antineoplastons in Texas. As a later court order would detail, Burzynski asked officials at the Texas Department of State Health Services if he could legally treat patients with antineoplastons.
The court order, which included a history of the steps he took in opening the clinic, states that the officials gave him a verbal green light, but never gave written consent.
Based on what the officials told him, he decided to open a private practice and research lab in a business park near the Westchase area.
The problem is that as he started treating patients with his therapy, he began achieving a number of remarkable successes.
I have to say in reviewing his case, his decision to go into private practice is likely what triggered everything that befell him. Had he “stayed in the fold” and continued his research, Pharma could have easily partnered with or bought him out, stole his therapies, or just kept him dependent on grants while also manipulating trials to make it seem anti-neoplastons was as or more effective than it was (its what they do).
At the risk of foreshadowing, like with ivermectin, Pharma eventually ended up doing the opposite – they conducted chart reviews and manipulated trials concluding that anti-neoplastons did not work. I believe they did this not only to “kill off” the popularity of the therapy, but also to support their unrelenting legal persecutions of Burzynski as you will read about below.
So basically, he opened a private clinic while separately starting a company that manufactured the anti-neoplastons, and then he treated cancer patients from all over. The treatment became increasingly popular by word-of-mouth via reports of increasing remissions. Whoa Nelly. In short, Burzynski was now “poking the bear.”
On the above point, to give you context for what happened (and to be able to read his Wikipedia page with sufficient skepticism for its veracity), know this:
Thomas Elias, the author of The Burzynski Breakthrough: The Most Promising Cancer Treatment and the Government’s Campaign to Squelch It. He remains convinced that the FDA persecuted Burzynski because he threatened the livelihood of the doctors on the agency’s review boards.
“These are the very people who stand to lose the most if Burzynski’s drug is proved,” he says. “These are the people who radiate kids’ brains at St. Jude‘s regularly…These are the people who are the enemies of this drug, because they have the most to lose from it…If this drug is approved, it will basically say that what they’ve been doing for all these decades is junk, and wasting money and lives.”
Elias says that he did not set out with the intent of writing a book hailing Burzynski, but the more he researched, the more the data pointed to one conclusion. He says he randomly selected patients throughout the country to interview and follow up on. Most of those who followed the treatment plan were alive and improving, he says. “But the ones who gave up on the treatment, they were dead. Without exception.”
I also submit the below, incredibly moving testimony by a Police Sergeant and father of a little girl with brain cancer who testified in support of Burzynski at a Congressional Hearing which was called in the late 1990’s to investigate the FDA’s repeated persecutions of Burzynski (umm, where is my Congressional hearing about the Medical Boards and the ABIM’s perrsecution of me and my colleagues for our advocacy for ivermectin in Covid? Ron my friend, are you reading this?). The father testified as follows:
“Kristin developed a highly malignant brain tumor that spread throughout her spine and her brain. The doctors told us that we had really two options, take her home, let her die, or bring her in for massive dosages of chemo and radiation simultaneously. In either event, she was going to die. They were quite certain of and very quickly. Believing her only chance to be the standard route, we gave her the chemo and radiation. It burnt her skull so bad, she had second-degree burns and her hair never came back. To change her diapers, we had to wear rubber gloves because her urine was so toxic and it burnt her. She still had cancer. We were told, Sorry. We’ve done everything we can. Now she’s going to die, probably within a couple of months.
And I conducted my own investigation into Dr. Burzynski. I have no doubt the man is not a fraud. I have no doubt that he does what he does out of earnest’s belief that his medicine works. Now you’re in a position to judge for yourself whether it works or not, but it’s well-established by the FDA that it’s non-toxic.
Eighteen months later, we took my daughter off the anti-oneuloplastin. She had not died. She had no signs of tumor. She remained free for 18 months of cancer. She died last July of neurological necrosis. Her brain fell apart from the radiation. The autopsy showed that she was completely cancer-free. Out of 52 cases of that disease ever, no one died cancer-free, just Chrissy. She didn’t die of a terminal illness. She died of my inability to care for her properly, and she died from bad advice. She died because there’s a government institution that disseminates false information and is not looking out for welfare of the people.
If you want to get emotionally riled up, watch and listen to him deliver the above testimony (I literally get tears welling up and then the feeling of wanting to punch a fucking wall comes over me every time I watch it):
The Regulatory And Legal “Lawfare” Crusade Against Dr. Stanislaw Burzynski
You need to know that almost all of the persecutory actions detailed below occurred before “the establishment” was able to “prove inefficacy” with fraudulent reviews and attempts at manipulating the studies. It was also before any patient complaints or lawsuits. The timing is the tell.
- In 1978, FDA representatives warned Burzynski that he was violating federal law because he was not administering antineoplastons in the context of a clinical trial. Burzynski promptly filed and then received Investigational New Drug (IND) status in 1979, allowing him to use it as an experimental therapy while studying it in clinical trials (15 points)
- In 1981, FDA wrote in a letter: “The FDA advises persons who inquire about Burzynski’s alleged cure that we do not believe the drug is fit for administration to humans and that there is no reason to believe Dr. Burzynski has discovered an effective cure for cancer (15 points)
- In 1983, the FDA commenced a civil action to try to close the clinic and stop all patients from receiving the medicine (25 points). Before the judge in this case had announced her ruling, the FDA sent her a letter warning her in advance.
- “If this court declines to grant the injunction sought by the government, thus permitting continued manufacture and distribution of anti-neoplastons, the government would then be obliged to pursue other less efficient remedies, such as actions for seizure, condemnation of the drugs, or criminal prosecution of individuals.” (25 points – insane)
- Ultimately the FDA obtained an injunction from the federal district court prohibiting Dr. Burzynski and the Burzynski Research Institute from shipping antineoplastons in interstate commerce without first obtaining the approval of the FDA. The injunction, however, did not preclude intrastate distribution of the antineoplastons (15 Points)
- In 1984, the Texas Board of Medical Examiners sent agents to try to convince his patients to file a complaint against him and his experimental treatment.
- Burzynski: “This was shocking me. What is surprising that they were using state money, taxpayer money, to travel long distances from Houston to California to convince my California patients to file complaints against me. This was completely irrational.” (20 points – insane).
- In July 1985, as part of a criminal investigation based on a referral from the FDA to the Department of Justice, the government applied for and obtained a warrant. FDA officials raided Burzynski’s office. They took whatever documents they could — including patient files — and dragged patients into grand jury hearings. Instead of securing indictments, the FDA only succeeded in making thousands of people feel their privacy had been violated (50 points)
- In 1988, the Texas Department of Health ordered Dr. Burzynski to cease and desist treating cancer patients with antineoplaston therapy absent FDA new drug or investigational drug approval (IND) (Problem – he already had IND status)
- In 1992, Burzynski was essentially forced to enter a lawsuit by one of his patients against Aetna Life Insurance company but it was dismissed despite brazen, illegal actions by Aetna and their lawyers (their only punishment was having to pay Burzynski’s legal fees). He appealed and won. From the appeal which he won, the judge summarizes what Aetna and their lawyers did to deserve the reprimand in the first case, it is worth a read. Note it is written by a Judge:
- Aetna, through the Hinshaw Firm, sent out a form letter to a large number of insurance companies. Aetna and Hinshaw describe the letter as an “informal discovery request.” That is a rather bland description. It opens: “This letter is sent to you as a result of an action filed by Aetna Life Insurance Company that may directly affect your company. You may have paid and may still be paying claims for cancer treatments of your insureds with an experimental substance used by Dr. Stanislaw Burzynski of Houston, Texas.” It next informs the recipient of the pending civil RICO action. Then appears the following sentence: “This letter is to warn you of potentially fraudulent claims for insurance reimbursement that may have been made to your company and to ask for your help in obtaining any claim history that your company may have with Dr. Burzynski” (emphasis supplied). The letter also contains several strongly pejorative statements. For example, it labels antineoplaston treatment as “worthless,” apprises the recipient of the 1983 FDA action against Burzynski and of unfavorable reports from the medical community on the treatment, notes that Dr. Burzynski has failed to receive FDA approval for the drug, and relays that a “Texas Grand Jury is currently investigating his operation.”
The judge who wrote the above granted that Burzynski’s appeal go forward.
- In May of 1993, the Texas Medical Board again took Burzynski to court. Listen to a different judge reflect on the case some years later:
“My memory isn’t quite clear what the Board’s problem was. The Board did not bring any expert witnesses to contest points that were raised by Dr. Bursyznski. Now, without an expert witness to render an opinion in certain areas, I can’t give any credence to an opinion raised by a layman. Some of the most dramatic testimony on Dr. Burzynski’s behalf came from Dr. Nicolas Petronas, a Georgetown University expert who was a member of the National Cancer Institute’s team that analyzed seven of Dr. Burzynski’s cases. The basic conclusion was that in five of the patients with brain tumors that were very large, the tumor resolved, disappeared. (50 points)
I am going to interrupt this exercise to show you a 42 second clip of a fiery and inspiring exchange between Burzynski and the Texas Board lawyer at that hearing.
Know that as a temper-prone, foul mouthed New Yorker who has himself “lost it” on the stand of late in a number of expert deposition defenses of persecuted doctors and patients during Covid, I “can identify” with his anger and words here, which were masterful really:
- 1993 – In response to the decision to find Burzynski not liable, the Texas Board then threatened the judge on Burzynski’s case that they would “re-write his proposal for his decision and proceed to take adverse action against Dr. Burzynski.” The judge advised them “that would not be a wise decision” (don’t mess with Texas judges apparently (15 points)
The FDA wouldn’t wrangle an indictment until 1995 — and that was a doozy: Seventy-five counts, mostly for mail fraud and shipping an unapproved drug across state lines, that would’ve put Burzynski behind bars for nearly 300 years.
It was a brazenly ridiculous, unfounded charge. Watch below as his lawyer Rick Jaffe explains the insanity of the above actions. Fun fact: Rick Jaffe was my lawyer as the lead plaintiff in a case we brought against the State of California for passing that insane Covid bill which mandated that California physicians only spout “consensus opinions” to their patients and not their own opinions):
- When the trial began in 1997, the jury deadlocked, and a mistrial was declared. The judge then tossed the 34 mail fraud counts, citing lack of evidence. For its part, the FDA dropped 40 counts, leaving — 12 years after the raid and seizure of patient records — only one count of contempt. Burzynski was promptly acquitted. (50 points)
Know that throughout the above proceedings, Burzynski’s patients picketed outside the courthouse and testified before Congress. From this article: “As far as they were concerned, the FDA was persecuting a noble man who merely wanted to offer a nontoxic alternative to radiation and chemotherapy.
It gets even better, because not only were his patients outraged but so were the jurors in his trial. From this article called “Twelve Angry Jurors” in 1997, some quotes:
The voir dire, where lawyers speak to prospective jurors in a group, seemed at times as if it would deteriorate into an angry mob scene. One prospective juror told prosecutors the FDA was like the Gestapo. A 24-year career marine said the case made him ashamed of his country, and that he found it very disturbing. When asked if they had any questions, one woman stood up and asked, “Why isn’t the FDA being prosecuted for violating our constitutional rights?” Jurors took just two-and-a-half hours to find Burzynski not guilty on the remaining charge.
- After the trial, an investigative arm of the Justice Department, the Office of Professional Responsibility, started investigating possible prosecutorial misconduct in the Burzynski case. After the not-guilty verdict, prosecutor Michael Clark was seen on local news shows, sweating profusely as he told reporters that he will be exonerated of any wrongdoing.
- Then, undeterred by the 1993 ruling, the Texas Medical Board took Dr. Burzynski to a higher district court. But get this, at the time, they claimed that “the efficacy of anti-neoplastons in the treatment of human cancers is not the issue” but rather that they wanted to suspend his license “because his treatments have never been FDA approved.” He was again exonerated (25 points)
Now, why would the Texas Medical Board continue on with this empty pursuit? Well, it was eventually realized, even by the mainstream press, that the Food and Drug Administration had been pressuring the Texas Medical Board to continue trying to take away Dr. Buryzinski’s medical license.
A clip from this TV news program that was featured in the documentary: “For this story, we wanted to talk to the FDA about its policies and procedures. The agency did agree to talk to us on background where it wouldn’t be quoted, but it repeatedly refused our request for on-camera interviews. While they were busy pressuring the Texas State Medical Board to try to revoke Dr. Burzynski’s medical license, they were even busier trying to revoke Dr. Burzynski completely from society by trying to place him in prison. And so the fiercest fight in FDA history began.
- In 2009, the FDA issued a warning letter to the Burzynski Research Institute, stating that an investigation had determined the Burzynski Institutional Review Board (IRB) “did not adhere to the applicable statutory requirements and FDA regulations governing the protection of human subjects.”
- In December 2010, the Texas State Board of Medical Examiners filed a multi-count complaint in the Texas State Office of Administrative Hearings against Burzynski for failure to meet state medical standards.
- In November 2012, a Texas State Office of Administrative Hearings administrative law judge ruled that Burzynski was not vicariously liable under Texas administrative law for the actions of staff at the clinic.
- In July 2014, the board filed a 202-page complaint against Burzynski to the Texas State Office of Administrative Hearings. The complaint addressed allegations by the Board including misleading patients into paying exorbitant charges, misrepresenting unlicensed persons to patients as licensed medical doctors
- In February 2017, following lengthy hearings, the Texas Medical Board recommended Burzynski’s medical license be revoked, with the revocation suspended, and a fine of $360,000 for billing irregularities and other violations
- Ed: know that “billing irregularities” are the achilles heel of every US physician. “They” can always find a miscoded claim when reviewing medical records, no matter how innocuous, and then they can charge the doctor with billing fraud and imprison them. Know this has happened to really good doctors I have come to know of. This is one of, if not the main reasons why my tele-health practice is fee-based and does not take insurance.
- In March 2017, the Texas Medical Board sanctioned Burzynski, placing him on probation and fining him $40,000. After being sanctioned for over 130 violations, he was allowed to keep his medical license and to continue to practice. Staff recommendations had been more punitive. Probation terms included additional medical training, disclosure of the Board’s ruling to patients and medical facilities, and monitoring of his patient records. (50 points)
1996 CONGRESSIONAL SUBCOMMITTEE HEARING IN SUPPORT OF DR. BURZYNSKI
Upon the commencement of the FDA’s 1995 grand jury against Dr. Burzynski, an oversight and investigation subcommittee was organized by Congressman Joe Barton in an attempt to intervene in the FDA’s relentless harassment of Dr. Burzynski and his patients.
Jurors from previous hearings and throngs of former patients showed up to protest. By this time Burzynski had saved the lives of more than 300 people who were supposed to have been dead.
Check out these comments from Chairman Joe Barton at the hearing (38 seconds)
Manipulation of Anti-Neoplaston Trials by the National Cancer Institute
From this account in 2011:
There was one minor ingredient in some of the antineoplastons that Burzynski had not patented because lawyers had counseled him that it was so common and well known that a patent would never be granted. The company took this ingredient to the National Cancer Institute (NCI), where the doctor who had defected from the Burzynski clinic was quickly made section chief.
They immediately received FDA approval for trials while Burzynski continued to be stonewalled in his attempts to receive FDA approval for trials. Burzynski had determined 12 years earlier that the ingredient was insignificant, and only needed to be included in one of the antineoplastons. He publicly predicted the trial would not work. Burzynski was frustrated by the bureaucratic corruption but with his background, not too surprised, he soldiered on.
When the National Cancer Institute’s research trials failed, associate director Michael Freidman came back to Burzynski and offered to do trials with his antineoplastons, but only if he would agree to make major changes to the protocols that he’d spent decades developing. At first he refused, and Freidman threatened to find other sources for antineoplastons. Burzynski fired back that federal employees shouldn’t contemplate patent infringement. The NCI finally agreed that Burzynski’s protocols would be followed. The protocols were simple and routine for cancer trials. Patients with very large tumors, multiple tumors, and metastases were to be excluded. These protocols are designed to rule out complicating factors that can skew results.
When a year passed with no patients being enrolled, Burzynski suspected something was amiss. The NCI said it had trouble finding patients, and then altered the protocols to include the complex cancers behind the back of Burzynski, who could have changed the instructions for dosages to treat such advanced cancers if he had known. The dosages would have had to have been increased at least three times. As the conflict escalated the NCI quit sending information to Burzynski who had to resort to legal means to get access to the trial data about his own medications.
He went on TV and said that he had gotten the distinct impression that the NCI wanted the patients to die so that the experimental trial would be over as soon as possible. When Dr. Burzynski finally got the data for the trial, he learned that there were only nine patients enrolled and they had suffered from severe fluid retention, something that he monitored and was able to prevent with his patients. He suspected that the antineoplastons were being diluted. The FDA later added insult to injury by publishing in a medical journal that the antineoplastons did not have cancer treatment potential. Some researcher made the mistake of including figures that showed the antineoplastons in the patients’ blood were three to 170 times less than the Burzynski clinic typically measured. The trial had been sabotaged. (100 points)
I then found this:
After all of the dust had settled, even more corruption came to light from internal memos. Going back all the way to 1991 the U.S. Department of Health and Human Services had been filing patents on the one ingredient Burzynski had not patented, claiming that the rights to manufacture, use, and license it belonged solely to the government. Shortly after the patents were filed, Michael Freidman, who had obstructed Burzynski’s trials at the National Cancer Institute, became deputy commissioner at the FDA, working directly under the commissioner who’d declared war on Burzynski. This breathtaking audacity did take Burzynski aback. Powerful government officials and the pharmaceutical industry had been conspiring to steal his invention.
DISINFORMATION CAMPAIGNS IN THE MEDIA
Recall that “Astroturfing” is when special interests disguise themselves and publish blogs, start Facebook and Twitter accounts, publish ads, letters to the editor, or simply post comments online to try to fool you into thinking that independent, authoritative, or grassroots movements are speaking.
Wikipedia is likely the most powerful tool in Disinformation and/or Astroturfing. Dr. Burzynski’s Wikipedia page is a huge, steaming pile of negative propaganda which portrays him as a fraud, profiteer, and a charlatan while completely discrediting antineoplaston therapy (40 points).
The below section from his Wikipedia page is particularly important as it, to me, represents a boilerplate example of how Pharma discredits effective therapies that are inconvenient to the industry. If you substitute ivermectin, HCQ, or chlorine dioxide for antineoplaston in the below paragraph, I can find dozens of news article using similar arguments and descriptors:
“Although Burzynski and his associates claim success in the use of antineoplaston combinations for the treatment of various diseases, and some of the clinic’s patients say they have been helped, there is no clinical evidence of the efficacy of these methods. The consensus among the professional community, as represented by the American Cancer Society[ and Cancer Research UK is that antineoplaston therapy is unproven, and the overall probability of the treatment turning out to be as claimed is low due to lack of credible mechanisms and the poor state of research after more than 35 years of investigation. Antineoplaston treatments have significant known side effects including severe neurotoxicity. Hypernatremia is also a significant risk given the high levels of sodium in antineoplaston infusions.”
Now you guys know why I have a tattoo on my right arm which says “Insufficient Evidence:”
Kory Scale Score for anti-neoplaston therapy: incalculable.
Hey readers, what therapy should be “run through the Kory Scale” next?
If you appreciate the effort and time I spend researching and writing my posts, Op-Ed’s and doctor defenses, support in the form of paid subscriptions is greatly appreciated.
Since we are on the topic of alternative cancer treatments, I would be remiss if I did not highlight my own complementary cancer care practice with my partner Scott Marsland, where we treat patients with combinations of scientifically supported repurposed medicines and dietary interventions based on the now validated Metabolic Theory Of Cancer as part of a prospective, observational study (which has just begun the data collection phase).
P.S. What I have seen in my almost one year of treating cancer patients with this approach are a growing minority of remarkable remissions, a majority of disease stability without progression, and another minority of non-responses, failures, and deaths (the latter outcome is something that others treating similarly ever seems to admit to on the internet). Cancer is a wicked disease but we are learning and we are helping many (but not all) immensely. I also believe that is what Dr. Burzynski (and now apparently his son) achieved with their treatment approaches.
I just want to say that writing this Substack is only one of my jobs, and I put a lot of work into it (at the cost of sleep and personal time). I believe in an ethos of paying people for their work but I do not require payment as I almost never place any content behind a paywall. If you love this Substack, get value out of it, and believe in paying people for their work, consider a paid subscription. Thanks, Pierre
Business
RFK Jr. says Hep B vaccine is linked to 1,135% higher autism rate
From LifeSiteNews
By Matt Lamb
They got rid of all the older children essentially and just had younger children who were too young to be diagnosed and they stratified that, stratified the data
The Centers for Disease Control and Prevention (CDC) found newborn babies who received the Hepatitis B vaccine had 1,135-percent higher autism rates than those who did not or received it later in life, Robert F. Kennedy Jr. told Tucker Carlson recently. However, the CDC practiced “trickery” in its studies on autism so as not to implicate vaccines, Kennedy said.
RFK Jr., who is the current Secretary of Health and Human Services, said the CDC buried the results by manipulating the data. Kennedy has pledged to find the causes of autism, with a particular focus on the role vaccines may play in the rise in rates in the past decades.
The Hepatitis B shot is required by nearly every state in the U.S. for children to attend school, day care, or both. The CDC recommends the jab for all babies at birth, regardless of whether their mother has Hep B, which is easily diagnosable and commonly spread through sexual activity, piercings, and tattoos.
“They kept the study secret and then they manipulated it through five different iterations to try to bury the link and we know how they did it – they got rid of all the older children essentially and just had younger children who were too young to be diagnosed and they stratified that, stratified the data,” Kennedy told Carlson for an episode of the commentator’s podcast. “And they did a lot of other tricks and all of those studies were the subject of those kind of that kind of trickery.”
But now, Kennedy said, the CDC will be conducting real and honest scientific research that follows the highest standards of evidence.
“We’re going to do real science,” Kennedy said. “We’re going to make the databases public for the first time.”
He said the CDC will be compiling records from variety of sources to allow researchers to do better studies on vaccines.
“We’re going to make this data available for independent scientists so everybody can look at it,” the HHS secretary said.
— Matt Lamb (@MattLamb22) July 1, 2025
Health and Human Services also said it has put out grant requests for scientists who want to study the issue further.
Kennedy reiterated that by September there will be some initial insights and further information will come within the next six months.
Carlson asked if the answers would “differ from status quo kind of thinking.”
“I think they will,” Kennedy said. He continued on to say that people “need to stop trusting the experts.”
“We were told at the beginning of COVID ‘don’t look at any data yourself, don’t do any investigation yourself, just trust the experts,”‘ he said.
In a democracy, Kennedy said, we have the “obligation” to “do our own research.”
“That’s the way it should be done,” Kennedy said.
He also reiterated that HHS will return to “gold standard science” and publish the results so everyone can review them.
International
RFK Jr. tells Tucker how Big Pharma uses ‘perverse incentives’ to get vaccines approved

From LifeSiteNews
By Matt Lamb
Kennedy defended his decision to fire all 17 members of the Advisory Committee on Immunization Practices, which he decried as a tool used to “rubber stamp” vaccines.
The vaccine approval process is a “bundle of perverse incentives” since pharmaceutical companies stand to make billions of dollars in revenue from it, Secretary of Health and Human Services Robert F. Kennedy Jr. told Tucker Carlson recently.
Kennedy appeared on Carlson’s show yesterday to discuss a variety of issues, including the potential link between autism and vaccines and his overhauling of the vaccine advisory committee at the Centers for Disease Control and Prevention last month.
Twenty years ago, Bobby Kennedy was exiled from polite society for suggesting a link between autism and vaccines. Now he’s a cabinet secretary, and still saying it.
(0:00) The Organized Opposition to RFK’s Mission
(6:46) Uncovering the Reason for Skyrocketing Rates of Autism… pic.twitter.com/g8T8te3kNC— Tucker Carlson (@TuckerCarlson) June 30, 2025
Kennedy began by explaining that Big Pharma has been targeting academic journals to ensure its products receive favorable reviews.
“The journals won’t publish anything critical of vaccines … there’s so much pressure on them. They’re funded by pharmaceutical companies, and they’ll lose advertising and revenue from reprints,” Kennedy said.
Kennedy then noted that Big Pharma will “pay to get something published in these journals,” before accusing industry leaders of pushing drugs on doctors and of hiring “mercenary scientists” to manipulate data until their product is deemed safe and effective.
The entire complex is broken due to the “perverse incentives,” he lamented.
Later in the interview, Kennedy defended his decision to fire all 17 members of the Advisory Committee on Immunization Practices (ACIP) in June, which he decried as a mere tool to “rubber stamp” vaccines.
It served as “a sock puppet for the industry that it was supposed to regulate,” Kennedy exclaimed, citing conflicts of interest for the overwhelming majority of its board members.
This sort of “agency capture” explains the lucrative nature of vaccines, he added.
— Matt Lamb (@MattLamb22) July 1, 2025
Kennedy then summarized the “perverse” process as follows:
First of all, the federal government often times actually designs the vaccine, [the National Institutes of Health] would design it, would hand it over to the pharmaceutical company. The pharmaceutical company then runs it … first through [the] FDA, then through [the] ACIP, and gets it recommended.
If you can get that recommendation you now got a billion dollars in — at least — revenues by the end of the year, every year, forever. So, there was a gold rush to add new vaccines to the schedule and ACIP never turned away a single vaccine … that came to them they recommended, and a lot of these vaccines are for diseases that are not even casually contagious.
Kennedy further pointed to the Hepatitis B shot for newborns as an example of how the industry has been corrupted.
In 1999, the CDC “looked at children who had received the hepatitis vaccine within the first 30 days of life and compared those children to children who had received the vaccine later — or not at all. And they found an 1,135% elevated risk of autism among the vaccinated children. It shocked them. They kept the study secret and manipulated it through five different iterations to try to bury the link,” he said.
“We want to protect public health,” Kennedy explained, but “these vaccines … can cause chronic disease, chronic injuries that last a lifetime.”
-
 Crime2 days ago
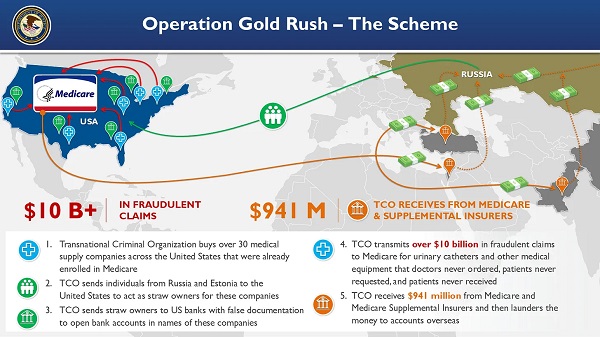
Crime2 days agoNational Health Care Fraud Takedown Results in 324 Defendants Charged in Connection with Over $14.6 Billion in Alleged Fraud
-

 Health2 days ago
Health2 days agoRFK Jr. Unloads Disturbing Vaccine Secrets on Tucker—And Surprises Everyone on Trump
-

 Business1 day ago
Business1 day agoElon Musk slams Trump’s ‘Big Beautiful Bill,’ calls for new political party
-

 Censorship Industrial Complex1 day ago
Censorship Industrial Complex1 day agoGlobal media alliance colluded with foreign nations to crush free speech in America: House report
-

 International23 hours ago
International23 hours agoCBS settles with Trump over doctored 60 Minutes Harris interview
-

 Business16 hours ago
Business16 hours agoLatest shakedown attempt by Canada Post underscores need for privatization
-

 Business15 hours ago
Business15 hours agoWhy it’s time to repeal the oil tanker ban on B.C.’s north coast
-

 Alberta15 hours ago
Alberta15 hours agoPierre Poilievre – Per Capita, Hardisty, Alberta Is the Most Important Little Town In Canada